Clone of Topical treatment of radiation-induced dermatitis

DRUGS IN CONTEXT
RIGOROUS – RAPID – RESPONSIVE
A continuous publication, open access, peer-reviewed journal
Topical treatment of radiation-induced dermatitis: current issues and potential solutions
Nicola Alessandro Iacovelli MD(1), Yvan Torrente MD, PhD(2,3), Adriana Ciuffreda MD(4), Vittorio A Guardamagna MD, PhD(5,6),Marta Gentili MS(7), Luca Giacomelli PhD(8,9), Paola Sacerdote PhD(10)
(1)Radiation Oncology Unit 2, Fondazione IRCCS Istituto Nazionale dei Tumori di Milano, Milan, Italy; (2)Stem Cell Laboratory, Department of Pathophysiology and Transplantation, Universitá degli Studi di Milano, Milan, Italy; (3)Unit of Neurology, Fondazione IRCCS Ca’ Granda Ospedale Maggiore Policlinico, Centro Dino Ferrari, Milan, Italy; (4)Medico Chirurgo, Specialista in Dermatologia e Venereologia, Dermatologia Pediatrica, Milan, Italy; (5)Division of Palliative Care and Pain Therapy, IRCCS Istituto Europeo di Oncologia IEO, Milan, Italy; (6)Director of ESMO, Designated Center of Integrated Oncology and Palliative Care, Milan, Italy; (7)Research Biologist, Private Practice, Milan, Italy; (8)Polistudium SRL, Milan, Italy; (9)Department of Surgical Sciences and Integrated Diagnostics, University of Genoa, Genoa, Italy; (10)Department of Pharmacological and Biomolecular Science, University of Milano, Milan, Italy
ABSTRACT
Approximately 95% of patients receiving radiotherapy (RT) will ultimately develop radiation-induced dermatitis (RID) during or after the course of treatment, with major consequences on quality of life and treatment outcomes. This paper reviews the pathophysiology of RID and currently used topical products for the prevention and treatment of RID. Although there is no consensus on the appropriate management, recent evidence suggests that the use of topical products supports to protect and promote tissue repair in patients with RID. Basic recommendations include advice to wear loose clothing, using electric razors if necessary, and avoiding cosmetic products, sun exposure or extreme temperatures. Based on mechanisms involved and on the clinical characteristics of oncological patients, the profile of the ideal topical product for addressing RID can be designed; it should have limited risk of adverse events, systemic adsorption and drug–drug interactions, should be characterized by multiple clinical activities, with a special focus on localized pain, and should have a careful formulation as some vehicles can block the RT beam.
Keywords: pain, quality of life, radiation-induced dermatitis, radiotherapy, skin toxicity, topical treatment.
CITATION
Iacovelli NA, Torrente Y, Ciuffreda A, Guardamagna AV, Gentili M, Giacomelli L, Sacerdote P. Topical treatment of radiation induced dermatitis: current issues and potential solutions. Drugs in Context 2020; 9: 2020-4-7. DOI: 10.7573/dic.2020-4-7
INTRODUCTION
Radiation-induced dermatitis (RID) is a very common side effect that is almost universally experienced by patients undergoing radiotherapy (RT) for cancer treatment. RID results from cutaneous or subcutaneous lesions due to external beam radiation. Indeed, it has been estimated that approximately 95% of patients receiving RT will ultimately develop RID during or after the course of treatment, with major consequences on quality of life and adherence to RT treatments, thereby affecting clinical outcomes.(1–3) However, at present, there is no consensus on the appropriate management of this condition. Therefore, there is urgent need for increased knowledge to guarantee a range of therapeutic options available for the treatment of RID.(1)
Recent evidence suggests that topical products may be used to protect and promote tissue repair in patients with RID, including within the prophylactic setting.(4,5)
The aim of this paper is to discuss current knowledge on RID and propose targets for the prevention/treatment of this condition. On these bases, the characteristics of the ‘ideal’ compound to address this side effect will be described.
Manuscripts for consideration in the present paper were retrieved via a PubMed search, using pertinent keywords (e.g. radiation-induced dermatitis). Papers were then selected for inclusion according to their relevance to the topic, as judged by the authors. The reference lists of the papers were also browsed to identify other suitable publications. Papers from personal collections of literature of the authors were also considered.
RID: BASIC CONCEPTS
Clinical manifestations
Key symptoms of RID include pain, ulceration, swelling, itching, burning, and physical and psychological discomfort. Opportunistic infections may also arise.(6–9) While these lesions may resolve over time, they can deeply influence quality of life and also limit the duration of treatment and the dose of radiation delivered, all of which have important consequences on therapy outcomes.(4,10)
The severity of RID is usually assessed by the physician according to standard instruments, such as the Common Terminology Criteria for Adverse Events (CTCAEs) scale (versions 2.0, 3.0, 4.0, or 4.03) or the Radiation Therapy Oncology Group/European Organization for Research and Treatment of Cancer (RTOG/EORTC) scale.(2) However, patient-reported outcome (PRO) tools (e.g. Skindex-16 or Brief Pain Inventory [BPI]) are sometimes used in clinical practice together with the CTCAE scale.(2) Up to 90% of patients will develop mild (grade 1) skin reactions, and approximately 20% of patients will develop severe forms of this condition.(11) Remarkably, concomitant systemic treatments (e.g. platinum-based regimens, cetuximab, 5-fluorouracil), administered to a good proportion of patients undergoing RT, can enhance the severity of RID.(2)
RID is characterized by an acute reaction when it occurs around the time of therapy and either chronic or late onset when it appears months or years after the end of treatment. Early skin reactions to RT usually occur within days to weeks from initiation, while late changes can happen even months or years after the completion of a RT course.
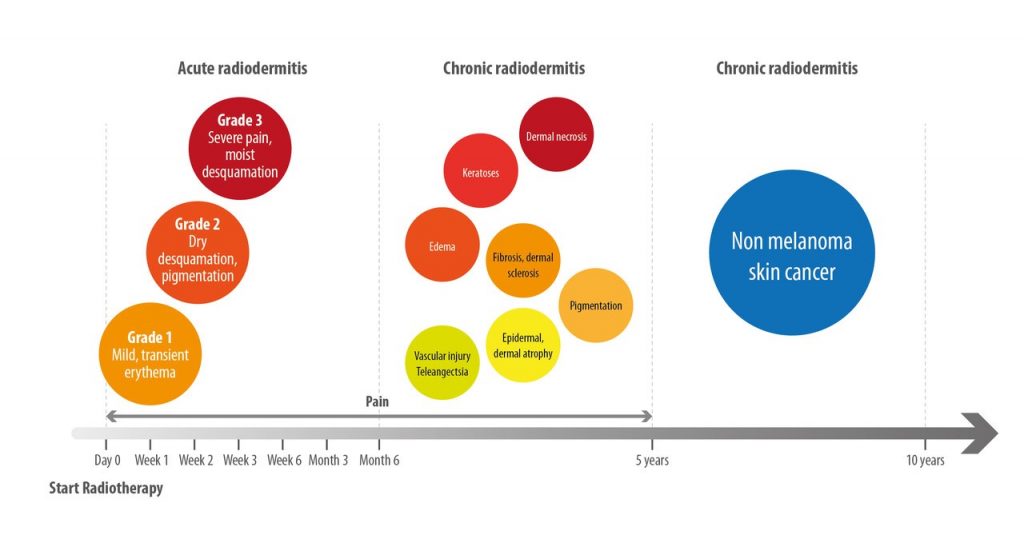
Usually, an acute phase of RID is defined by skin lesions that appear within 90 days from the start of treatment. Injury can first manifest as a transient erythema that may be reported as early as within the first 24 hours after the initiation of RT and is resolved within a few days (Figure 1).(12,13) Later, during weeks 2–4 of therapy, a more sustained generalized erythema and the onset of pain may be reported often associated with other skin changes, such as dryness and hyperpigmentation. During weeks 3–6 (from the start of therapy), dry desquamation associated with pain, pruritus, and scaling can develop if the cumulative radiation dose reaches 20 Gy, and moist desquamation may occur if total radiation dose to the skin is ≥40 Gy.(14) In this latter case, temporary interruption of RT, until the area is able to re-epithelialize, may be necessary.(15)
Chronic skin reactions to RT develop more than 90 days after the completion of radiation treatment.(12) These reactions include vascular injury resulting in telangiectasia, epidermal thinning, dermal atrophy, pigmentation, fibrosis, edema, keratosis, and, even, dermal necrosis. Moreover, patients show increased risk for the development of non-melanotic cutaneous malignancies in the course of their life.
Pathophysiology
Injury from RT derives from the generation of free radicals and reactive oxygen species in the rapidly dividing cells of the basal layer and underlying dermis.(12) Within a few hours from the start of treatment, this action decreases the number of functional stem cells, induces changes in endothelial cells, and promotes inflammation by the release of several cytokines and chemokines, most notably interleukin (IL)-1α, IL-1β, tumor necrosis factor-α, IL-6, IL-8, C–C motif chemokine ligand 4 (CCL4), C–X–C motif chemokine ligand 10 (CXCL10), and C–C motif chemokine ligand 2 (CCL2).15,16 Over the subsequent 2–4 weeks, a sustained generalized erythema develops, most likely associated with local degeneration and edema, resulting from infiltration of leukocytes into the irradiated skin, which is a ‘hallmark’ of radiation-induced skin injury. Dryness and epilation, often observed at this time of the course of RID, result from the destruction of sebaceous glands and hair follicles.(17–19)
Moreover, differing from other forms of mechanical or chemical damage to the skin, RT-induced damage is repetitive and accumulates over the course of treatment, leading also to a delayed disruption of the epidermal barrier. Indeed, chronic effects induced by RT comprise changes of the vasculature and connective tissue of the cutaneous and subcutaneous layers. Remodeling of epithelium may then lead to the formation of telangiectasias.(20) With accumulating doses of radiation, if the healing process based on growth factors, such as fibroblast growth factor (FGF) or epidermal growth factor (EGF), is impaired,(21) melanocytes and fibroblasts can be destroyed, causing hypopigmentation and atrophy of the derma.(12)
EPIDEMIOLOGY OF RID
RID is a widespread, although often neglected, side effect of RT. Indeed, at least 50% of patients with any type of cancer receive some form of RT during their disease history.(4) This prevalence is even higher in patients with head and neck and breast cancer: it has been estimated that 80–90% of all patients with head and neck cancer receiving RT develop RID, and 25% develop severe skin reactions.(22) In the breast cancer setting, one of the most widespread forms of cancer worldwide, 45% of patients receive RT, and the wide majority of them (74–100%) report RID.(23) Furthermore, chronic RID develops in one out of three patients and can appear up to 10 years after the completion of RT.(24) As >80% of all women treated for breast cancer survive for ≥10 years, the burden of chronic RID is mounting.(25) The risk of developing RID depends on various treatment-related factors: the radiation dose during a single delivery, the total dose, the duration of exposure, the volume of the treated area, and the combination with other therapies, such as chemotherapy.(26) Furthermore, RID can be influenced by patient-related factors that include high body mass index (BMI), smoking, nutritional status, pre-existing skin diseases, such as psoriasis, and genetic susceptibility.
Skin damage has also been associated with chemotherapy, although incidence of this event has not been precisely estimated.(26) Of note, radiation recall dermatitis is an acute inflammatory skin reaction occurring in a skin area previously exposed to RT and triggered by subsequent intake of a drug, most commonly a chemotherapeutic agent. The incidence of radiation recall dermatitis cannot be precisely determined, as most evidence comes from case report studies. Nevertheless, the effect has been reported in association with the use of several antineoplastic drugs.(27)
THE RATIONALE AND BASES FOR MANAGEMENT OF RID
As of today, management of RID is still based on preliminary evidence.(4) Indeed, at present, it is not possible to propose any firm recommendation to prevent or reduce RID.(2,4,20,28,29)
However, some basic recommendations can be made, according to the Multinational Association of Supportive Care in Cancer (MASCC) guidelines(20): patients should be advised to wear loose clothing, use electric razors (if necessary), and avoid cosmetic products, sun exposure or extreme temperatures.(12) Moreover, regular skin care assessment is mandatory during RT treatments.
In multimorbid, often polytreated and frail patients, like many in the oncological population, topical substances may represent the best initial option for treatment of skin toxicity.(1,30–32)
It is commonly accepted that daily topical treatment could be useful if started right from the beginning of RT to prevent the onset of RID.(4)
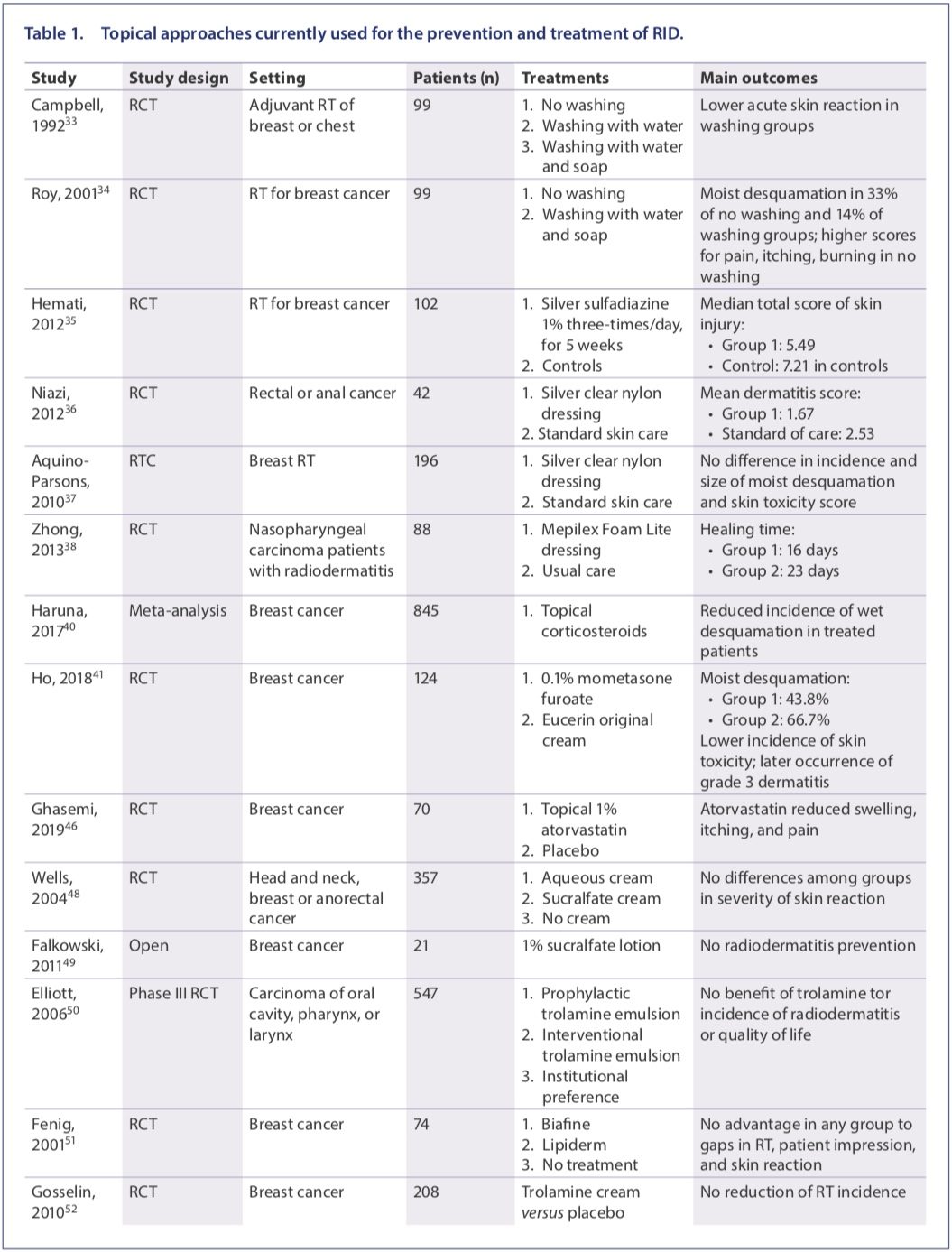
Available information on currently used topical approaches for the prevention and treatment of RID is presented in Table 1, while an overview of those approaches is presented in the following section.
DIFFERENT TREATMENTS FOR RID: AN OVERVIEW
Washing
Following standard hygiene practices is undoubtedly the most advantageous choice in the management of RID. Two wellconducted randomized controlled trials (RCTs) showed that washing with mild soap and lukewarm water reduces itching and RID severity.(33,34)
Dressings
Silver sulfadiazine, a topical antibacterial, is used as cream for second- and third-degree burns. This molecule is also endowed with anti inflammatory properties and barrierenhancing functions, thereby contributing to protect the skin from infectious agents.(1) In a preliminary study, the use of silver sulfadiazine–containing dressing was associated with a lower total severity of RID.(35) Similar findings were reported for silver nylon dressing(36,37) or for Mepilex Foam Lite dressing.(38)
Laser therapy
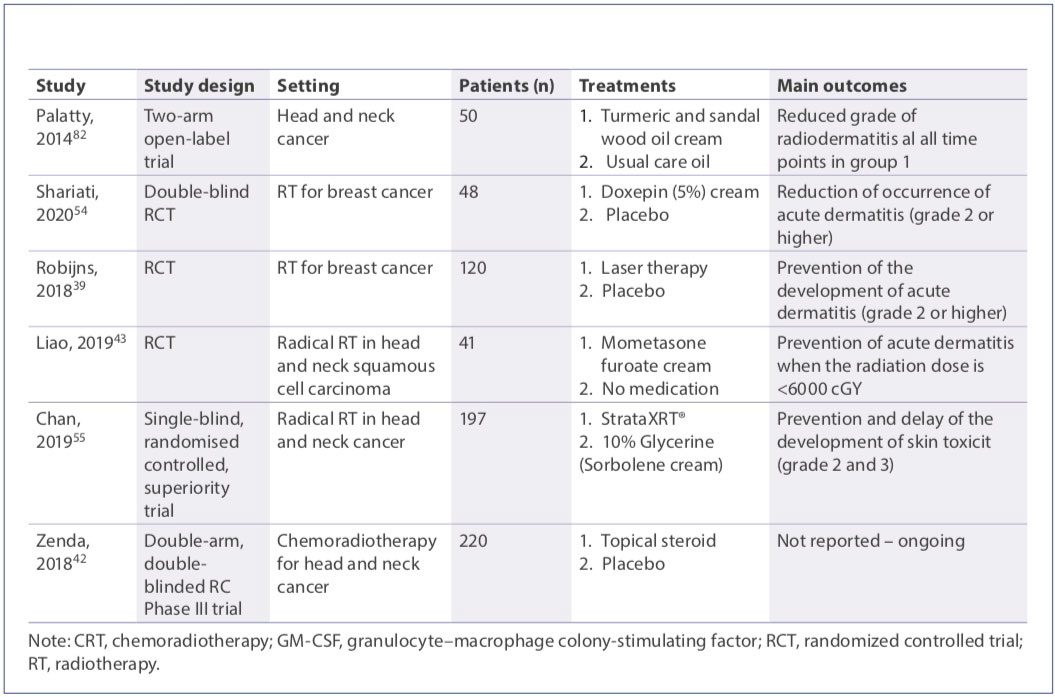
Photobiomodulation therapy after the RT session twice a week was tested in breast cancer patients. The effectiveness of the treatment was evaluated by a quality-of-life questionnaire and results in a beneficial effect.(39)
Pharmaceuticals and biological agents
Topical corticosteroids are commonly prescribed for RID because of their ability to counteract the radiation-induced release of cytokines.(32) While the efficacy of this therapeutic strategy is established in breast cancer(40,41) and is currently under evaluation in head and neck cancer,(42,43) the use of topical corticosteroids is frequently associated with the onset of clinically relevant adverse events.(44) In particular, the prolonged use of steroids leads to skin thinning, which may be particularly contraindicated in this condition. Statins display some antiinflammatory, immunomodulatory, antioxidant, metabolic, and antibacterial properties and can have some efficacy in the reduction of severity of skin disorders, such as psoriasis, dermatitis, uremic pruritus, and vitiligo.(45) In a preliminary experience, Ghasemi and colleagues investigated the topical use of atorvastatin.(46)
In topical formulation, sucralfate presents barrier abilities, antibacterial activity, anti-inflammatory effects, and angiogenesis-promoting capabilities.(47) However, sucralfate did not reduce the severity of RID or improve patient-reported symptoms.(48,49)
Trolamine is extensively used for the management of RID, as it is supposed to function as a non-steroidal anti-inflammatory molecule.(1) However, several RCTs did not show any advantage for trolamine over supportive care or even placebo in treating RD.(50–53) Doxepin cream has been recently shown to prevent dermatitis when applied after RT in breast cancer patients.(54)
Silicone-based agents in vitro have been shown to regulate fibrosis and wound healing, although with potential practical inconvenience due to non-perfect adherence to the skin when bathing and in men with facial hair. StrataXRT®, a silicone-based film-forming agent, was shown to be effective in delaying the development of skin toxicity.(55)
EGF stimulates the proliferation of human epidermal stem cells, fibroblasts, and keratinocytes, thus contributing to skin lesion healing.(56) Kang and colleagues suggested that topical EGF leads to a diminished incidence of toxicity when compared with historical data.(57)
Granulocyte–macrophage colony-stimulating factor (GM–CSF) promotes macrophage maturation and activity. When compared with patients receiving topical steroids alone, patients on topical steroids and GM-CSF-soaked gauze displayed reduced severity of RID and milder pain.(58)
Non-pharmaceutical agents
Several non-pharmaceutical agents have been proposed for the amelioration of RID. Here, we review the available topical treatments and one oral agent. The agents have been classified according to their main property. This does not exclude that one agent could present more than only one feature.
Anti-inflammatory agents
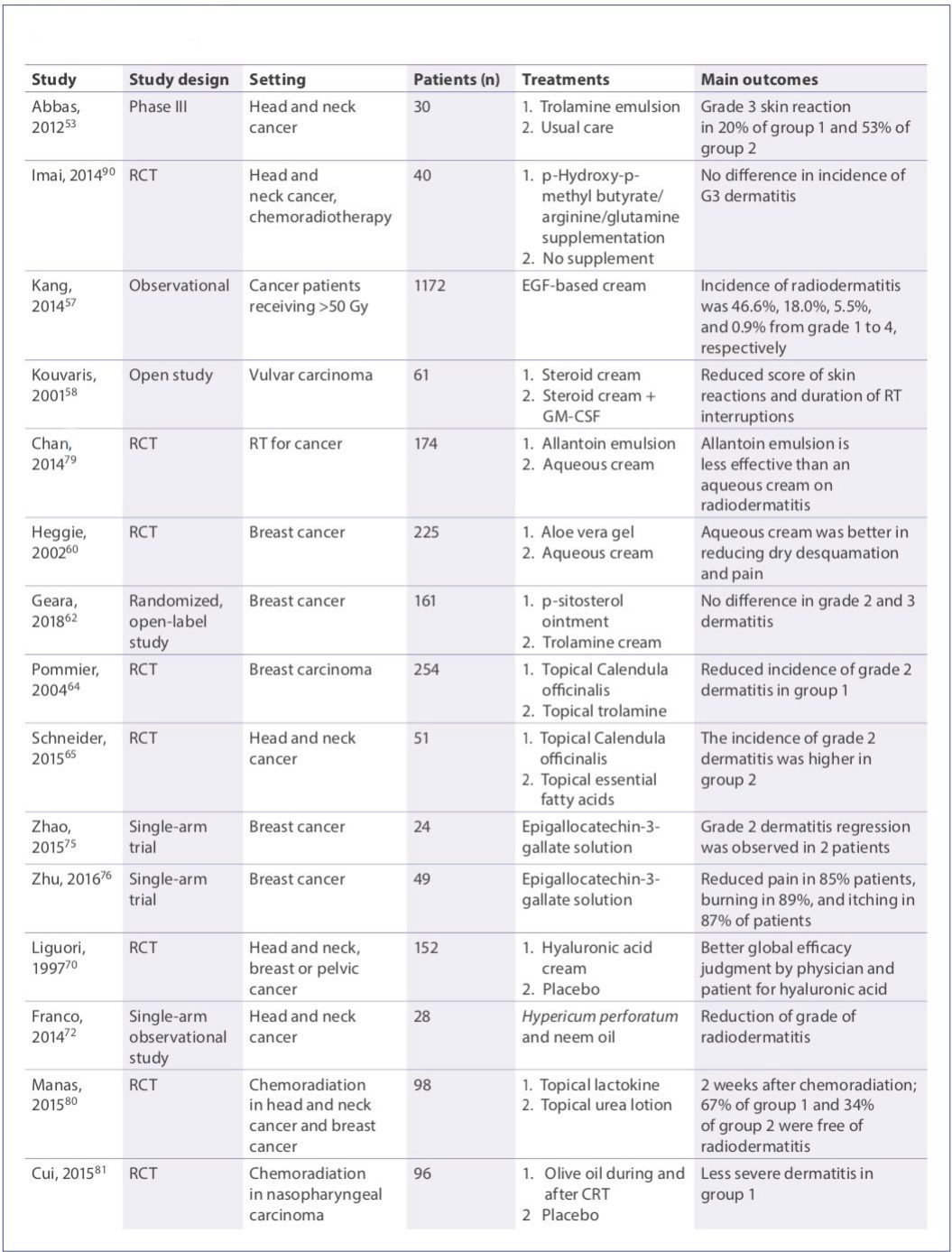
- Aloe vera is an anti-inflammatory herbal therapy endowed with protective and healing abilities.(59) However, despite these promising characteristics, aloe vera does not reduce the severity of RID.(60)
- Beta-sitosterol is a herbal formulation presenting antibacterial, analgesic, and anti-inflammatory effects.(61) In a recent clinical trial, beta sitosterol and trolamine were not associated with relevant improvements in RID, although the incidence of severe pruritus and local pain were both significantly reduced with beta-sitosterol.(62)
- Calendula officinalis has anti-inflammatory, antibacterial, antifungal, antioxidant, and angiogenic abilities.(63) In an RCT, calendula significantly lowered the frequency of grade ≥2 acute dermatitis compared with trolamine (41 versus 63%, p<0.001). Moreover, patients assigned to calendula required fewer interruptions in RT and reported milder pain.(64) These findings were confirmed in a more recent pilot randomized trial.(65)
- Hyaluronic acid (HA) is widely used in wound dressings, skin substitute products, and other applications in the field of regenerative medicine and dermatology.(66,67) HA is used in high-molecular weight and low-molecular weight forms, and the use of conjugated high- and lowmolecular weight HA can further increase the efficacy of this compound, in a synergic fashion.(68) In vitro, HA protected fibroblasts from radiation damage,(69) and in a double-blind RCT, HA was able to reduce the incidence of severe RID in 152 patients undergoing RT for head and neck, breast, or pelvic carcinomas.(70) Recent evidence also supports the clinical use of HA for topical administration in patients with inflammatory conditions at the level of mucosa.(71)
- In a pilot study, Franco and colleagues prospectively evaluated the role of Hypericum perforatum and neem oil in the treatment of acute skin toxicity.(72) This approach was preliminary and suggested to be safe and active in the management of this condition.
- Glutamine has been proposed to prevent oral RID and mucositis. However, a study evaluating the efficacy of oral glutamine showed no significant effect on the severity of oral mucositis.(73)
Antioxidant agents
- Catechins, in particular epigallocatechin-3-gallate (EGCG), present antioxidant activities that may promote healing of skin damage caused by exposure to ultraviolet light.(74) In a phase I/II clinical trial, topical EGCG showed several beneficial effects.(75,76)
- Vitamins have also been tested in this setting. Ascorbic acid is endowed with powerful antioxidant and free radical-scavenging abilities. These characteristics led Halperin and colleagues to study the possible protective role of ascorbic acid in RID, but their study failed to demonstrate any benefit of topical ascorbic acid in this setting.(77) Pantothenic acid (vitamin B5) has a central role in metabolism, and is essential for the integrity of normal skin. However, when compared with no treatment, a topical cream with pantothenic acid failed to show an enhanced protective effect against RID.(78)
- Topical preparations that contain comfrey extract, which comprises allantoin, may have some applications in the treatment of skin irritation. Indeed, in a doubleblind trial, patients assigned to the allantoin-containing cream had a lower severity of pain and itching at week 3 compared with those on aqueous cream.(79)
- Lactokine is a protein derived from milk. In a pilot study, topical administration of a lactokine-containing product was effective in preventing and reducing the grade of RID in head and neck and breast cancer patients.(80)
- In an RCT on 94 patients with head and neck cancer, the acute RID that resulted was decreased in intensity with the application of olive oil.(81) In another small randomized study on 50 patients, a cream containing turmeric and sandal wood oil was able to prevent the onset of RID, but the results require further validation in larger doubleblind trials.(82)
FROM BENCH TO BEDSIDE: HOW TO IMPROVE TREATMENT OF RID
RID has a major impact on quality of life of cancer patients and may interfere with oncologic therapies, thus reducing their effect.(4) In particular, pain is perceived as among the most severe symptoms associated with RID,(7) and also other symptoms experienced by patients – for example, itching and burning – are bothersome for patients.
From a basic research perspective, an improved understanding of the mechanisms of radiation-induced damage of the skin will lead to the opportunity to better schedule both preventive and curative strategies. Moreover, a large effort is needed to find predictive factors for the onset and severity of RID, also at a genomic level.(2)
It is of utmost importance to conduct proper epidemiological studies and nationwide surveys, with the aim to capture a clearer picture of incidence of RID and treatment approaches in different centers (e.g. primary versus referral centers).
Box 1. Practical tips for the clinical management of RID.
- Wash irradiated skin with lukewarm water and a mild pH, neutral or non-alkaline, soap daily
- Shave with a sharp, disinfected wet razor or with non-traumatizing electric razor
- Wear loose-fitting clothes in order to prevent friction injuries over the irradiated area
- Avoid extreme temperatures and sun exposure
- Avoid the use of metallic-based topical products, perfumes, and the use of tapes and adhesives
- Choose a proper topical product for each patient in order to keep the skin hydrated
- Any topical product should not be applied over the irradiated skin from 1 to 4 hours before the daily treatment
- Consider dressings and advanced medications in case of wet desquamation
- Prevent superinfections
- Consider topical or systemic antimicrobials in case of infection
- In case of grade 3 RID, every effort should be made to try not to interrupt RT
- In case of grade 4 RID, consider discontinuation of RT
RID, radiation-induced dermatitis; RT, radiation therapy.
At present, there is no standard treatment for RID, and a more rational approach to the therapy of this condition has been advocated.(4) Properly designed prospective studies, possibly RCTs, investigating RID should be conducted to collect highlevel evidence on this topic, which is currently lacking. These studies should contemplate PROs and quality-of-life measures as main outcome measures. An important pitfall is the lack of a reliable objective tool to measure RID. Methods used and reported in the literature are reflectance spectrophotometry, a non invasive technique aimed at measuring cellular pigments, and trans-epidermal water loss (TEWL) measurement, to analyze the epidermal barrier function.(83,84)
To overcome current doubts and help clinicians in managing patients with RID, here we provide some practical tips that can be easily suggested to patients (Box 1).
The ideal therapy for RID should present a number of characteristics and be based upon a rational selection of components. First, cancer patients are in almost all cases polymedicated, and receiving a number of treatments associated with bothersome adverse events. Hence, a topical product for the therapy of RID, with limited risk of systemic adsorption and drug–drug interaction, may be preferred, in line with previous recommendations in the dermatological setting.(30) Moreover, the product should address the complex array of manifestations of RID, and hence should be characterized by the combinations of a few selected active principles with different mechanisms of actions or targets and multiple clinical activities to counteract such diverse symptoms. Agents able to prevent the earlier events, such as production of free oxygen radicals or inflammatory mediators, should be combined with molecules that could stimulate healing and regeneration. In particular, as pain is recognized to be the most bothersome symptom associated with RID, particular attention should be paid to its management. The investigation in this setting of molecules with an established action on localized pain (e.g. carnitine85,86) can be of the highest interest.
The need of multiple actions should be, however, weighed against the need for limiting risk of adverse events and drug–drug interactions, and therefore the minimum possible number of components should be included. Furthermore, the formulation should be carefully selected: for instance, an oily phase may block the penetration of the RT beam. Hence, formulations without an oily phase, such as gels, may be preferred over those containing this (e.g. creams).
Proper selection of vehicle has also a major role, as it may influence the penetration into the different skin layers, and thereafter the cell targets eventually reached. Liposome formulations are able to uniformly penetrate the horny layer leading to a 4–14-times higher local concentration of active substances compared with conventional emulsions.(87) Moreover, beyond their role as carriers, liposomes can be considered as active agents, given their richness in phospholipids and omega fatty acids, for the treatment of dry, scaly, and flaky skin.(87) In an in vitro study, an ascorbate phosphatidylcholine liposome was shown to overcome the stratum corneum and deliver the active agent into the dermis to prevent photodamage.(88) These findings were mirrored in another experimental study.(89)
Hence, a topical product containing rationally selected components able to address pain, skin damage, and the other bothersome symptoms of RID – delivered by proper vehicle – ensuring, at the same time, optimal chemo-physical properties would be of the highest interest in the current treatment scenario for RID. Such a compound may be used also in the preventive setting, in line with recent suggestions,(4) in order to limit the onset of RID, improve the quality of life, and reduce the risk of RT interruptions.
Contributions: All authors contributed equally to the preparation of this review. All named authors meet the International Committee of
Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given
their approval for this version to be published.
Disclosure and potential conflicts of interest: LG is the owner of Polistudium, the consultancy agency involved in
the editorial assistance of the present paper. The other authors declare that they have no conflicts of interest. The International
Committee of Medical Journal Editors (ICMJE) Potential Conflicts of Interests form for the authors is available for download at: https://www.drugsincontext.com/wp-content/uploads/2020/05/dic.2020-4-7-COI.pdf
Acknowledgements: Editorial assistance was provided by Aashni Shah, Barbara Bartolini, and Laura Brogelli, MD (Polistudium, Milan, Italy).
Funding declaration: Editorial assistance was supported by Welcare Industries.
Copyright: All Copyright © 2020 Iacovelli NA, Torrente Y, Ciuffreda A, Guardamagna AV, Gentili M, Giacomelli L, Sacerdote P. Published by Drugs in Context under Creative Commons License Deed CC BY NC ND 4.0 which allows anyone to copy, distribute, and transmit the article provided it is properly attributed in the manner specified below. No commercial use without permission.
Correct attribution: All Copyright © 2020 Iacovelli NA, Torrente Y, Ciuffreda A, Guardamagna AV, Gentili M, Giacomelli L, Sacerdote P. https://doi.org/10.7573/dic.2020-4-7 Published by Drugs in Context under Creative Commons License Deed CC BY NC ND 4.0.
Correspondence: Luca Giacomelli, Polistudium SRL, Milan, Italy. – luca.giacomelli@polistudium.it
Provenance: submitted; externally peer reviewed.
Submitted: 14 April 2020; Peer review comments to author: 4 May 2020; Revised manuscript received: 15 May 2020;
Accepted: 18 May 2020; Publication date: 12 June 2020.
Drugs in Context is published by BioExcel Publishing Ltd. Registered office: Plaza Building, Lee High Road, London, England, SE13 5PT.
BioExcel Publishing Limited is registered in England Number 10038393. VAT GB 252 7720 07.
For all manuscript and submissions enquiries, contact the Editor-in-Chief – gordon.mallarkey@bioexcelpublishing.com
For all permissions, rights and reprints, contact David Hughes – david.hughes@bioexcelpublishing.com
References
- Rosenthal A, Israilevich R, Moy R. Management of acute radiation dermatitis: a review of the literature and proposal for treatment algorithm. J Am Acad Dermatol. 2019;81(2):558–567. https://doi.org/10.1016/j.jaad.2019.02.047
- Iacovelli NA, Galaverni M, Cavallo A, et al. Prevention and treatment of radiation-induced acute dermatitis in head and neck cancer patients: a systematic review. Future Oncol. 2018;14(3):291–305. https://doi.org/10.2217/fon-2017-0359
- Russi EG, Moretto F, Rampino M, et al. Acute skin toxicity management in head and neck cancer patients treated with radiotherapy and chemotherapy or EGFR inhibitors: literature review and consensus. Crit Rev Oncol Hematol. 2015;96(1):167-182. https://doi.org/10.1016/j.critrevonc.2015.06.001
- Seité S, Bensadoun RJ, Mazer JM. Prevention and treatment of acute and chronic radiodermatitis. Breast Cancer (Dove Med Press). 2017;9:551–557. https://doi.org/10.2147/BCTT.S149752
- Matovina C, Birkeland AC, Zick S, et al. Integrative medicine in head and neck cancer. Otolaryngol Head Neck Surg. 2017;156(2): 228–237. https://doi.org/10.1177/0194599816671885
- Schnur JB, Love B, Scheckner BL, et al. A systematic review of patient-rated measures of radiodermatitis in breast cancer radiotherapy. Am J Clin Oncol. 2011;34(5):529–536.
- Presta G, Puliatti A, Bonetti L, et al. Effectiveness of hyaluronic acid gel (Jalosome soothing gel) for the treatment of radiodermatitis in a patient receiving head and neck radiotherapy associated with cetuximab: a case report and review. Int Wound J. 2019;16(6):1433–1439. https://doi.org/10.1111/iwj.13210
- Beamer LC, Grant M. Using the Dermatology Life Quality Index to assess how breast radiodermatitis affects patients’ quality of life. Breast Cancer (Auckl). 2019;13:1178223419835547. https://doi.org/10.1177/1178223419835547
- Sutherland AE, Bennett NC, Herst PM. Psychological stress affects the severity of radiation-induced acute skin reactions in breast cancer patients. Eur J Cancer Care (Engl). 2017;26(6). https://doi.org/10.1111/ecc.12737
- Kodiyan J, Amber KT. Topical antioxidants in radiodermatitis: a clinical review. Int J Palliat Nurs. 2015;21(9):446–452. https://doi.org/10.12968/ijpn.2015.21.9.446
- Chan RJ, Larsen E, Chan P. Re-examining the evidence in radiation dermatitis management literature: an overview and a critical appraisal of systematic reviews. Int J Radiat Oncol Biol Phys. 2012;84(3):e357–e362. https://doi.org/10.1016/j.ijrobp.2012.05.009
- Hegedus F, Mathew LM, Schwartz RA. Radiation dermatitis: an overview. Int J Dermatol. 2017;56(9):909–914. https://doi.org/10.1111/ijd.13371
- Bray FN, Simmons BJ, Wolfson AH, Nouri K. Acute and chronic cutaneous reactions to ionizing radiation therapy. Dermatol Ther (Heidelb). 2016;6(2):185–206. https://doi.org/10.1007/s13555-016-0120-y
- McQuestion M. Evidence-based skin care management in radiation therapy: clinical update. Semin Oncol Nurs. 2011;27:e1–e17. https://doi.org/10.1016/j.soncn.2011.02.009
- Iacovelli NA, Naimo S, Bonfantini F, et al. Preemptive treatment with Xonrid®, a medical device to reduce radiation induced dermatitis in head and neck cancer patients receiving curative treatment: a pilot study. Support Care Cancer. 2017;25(6):1787–1795. https://doi.org/10.1007/s00520-017-3569-z
- Wickline MM. Prevention and treatment of acute radiation dermatitis: a literature review. Oncol Nurs Forum. 2004;31:237–247.
https://doi.org/10.1188/04.ONF.237-247 - de Pauli Paglioni M, Alves CGB, Fontes EK, et al. Is photobiomodulation therapy effective in reducing pain caused by toxicities related to head and neck cancer treatment? A systematic review. Support Care Cancer. 2019;27(11):4043–4054. https://doi.org/10.1007/s00520-019-04939-2
- Glover D, Harmer V. Radiotherapy-induced skin reactions: assessment and management. Br J Nurs. 2014;23(4):S28, S30–S35.
https://doi.org/10.12968/bjon.2014.23.Sup2.S28 - Ryan JL. Ionizing radiation: the good, the bad, and the ugly. J Invest Dermatol. 2012; 132(3 Pt 2):985–993. https://doi.org/10.1038/jid.2011.411
- Wong RK, Bensadoun RJ, Boers-Doets CB, et al. Clinical practice guidelines for the prevention and treatment of acute and late radiation reactions from the MASCC Skin Toxicity Study Group. Support Care Cancer. 2013;21(10):2933–2948. https://doi.org/10.1007/s00520-013-1896-2
- Kong M, Hong SE. Topical use of recombinant human epidermal growth factor (EGF)-based cream to prevent radiation dermatitis in breast cancer patients: a single-blind randomized preliminary study. Asian Pac J Cancer Prev. 2013;14(8):4859–4864. https://doi.org/10.7314/APJCP.2013.14.8.4859
- Ferreira EB, Vasques CI, Gadia R, et al. Topical interventions to prevent acute radiation dermatitis in head and neck cancer patients: a systematic review. Support Care Cancer. 2017;25(3):1001–1011. https://doi.org/10.1007/s00520-016-3521-7
- Schnur JB, Ouellette SC, Dilorenzo TA, et al. A qualitative analysis of acute skin toxicity among breast cancer radiotherapy patients. Psychooncology. 2011;20(3):260–268. https://doi.org/10.1002/pon.1734
- Whelan TJ, Pignol JP, Levine MN, et al. Long-term results of hypofractionated radiation therapy for breast cancer. N Engl J Med. 2010;362(6):513–520. https://doi.org/10.1056/NEJMoa0906260
- American Cancer Society. Cancer Facts & Figures 2017. Atlanta, GA: American Cancer Society; 2017. https://www.cancer.org/. Accessed October 12, 2017.
- Sibaud V, Leboeuf NR, Roche H, et al. Dermatological adverse events with taxane chemotherapy. Eur J Dermatol. 2016;26(5): 427–443. https://doi.org/10.1684/ejd.2016.2833
- Burris HA 3rd, Hurtig J. Radiation recall with anticancer agents. Oncologist. 2010;15(11):1227–1237. https://doi.org/10.1634/theoncologist.2009-0090
- Chan RJ, Webster J, Chung B, et al. Prevention and treatment of acute radiation-induced skin reactions: a systematic review and meta-analysis of randomized controlled trials. BMC Cancer. 2014;14:53. https://doi.org/10.1186/1471-2407-14-53
- Greenlee H, DuPont-Reyes MJ, Balneaves LG, et al. Clinical practice guidelines on the evidence-based use of integrative therapies during and following breast cancer treatment. CA Cancer J Clin. 2017;67(3):194–232. https://doi.org/10.3322/caac.21397
- Pereira MP, Ständer S. Therapy for pruritus in the elderly: a review of treatment developments. Expert Opin Pharmacother. 2018;19(5):443–450. https://doi.org/10.1080/14656566.2018.1444752
- Haas ML, Moore-Higgs GJ. Principles of Skin Care and the Oncology Patient. Pittsburgh, PA: Oncology Nursing Society; 2010.
- Hymes SR, Strom EA, Fife C. Radiation dermatitis: clinical presentation, pathophysiology, and treatment 2006. J Am Acad Dermatol. 2006;54:28–46. https://doi.org/10.1016/j.jaad.2005.08.054
- Campbell IR, Illingworth MH. Can patients wash during radiotherapy to the breast or chest wall? A randomized controlled trial. Clin Oncol. 1992;4(2):78–82. https://doi.org/10.1016/S0936-6555(05)80971-9
- Roy I, Fortin A, Larochelle M. The impact of skin washing with water and soap during breast irradiation: a randomized study. Radiother Oncol. 2001;58(3):333–339. https://doi.org/10.1016/S0167-8140(00)00322-4
- Hemati S, Asnaashari O, Sarvizadeh M, et al. Topical silver sulfadiazine for the prevention of acute dermatitis during irradiation for breast cancer. Support Care Cancer. 2012;20(8):1613–1618. https://doi.org/10.1007/s00520-011-1250-5
- Niazi T, Vuong T, Azoulay L, et al. Silver clear nylon dressing is effective in preventing radiation induced dermatitis in patients with gastrointestinal cancer: results from a phase III study. Int J Radiat Oncol Biol Phys. 2011;81(2):S171. https://doi.org/10.1016/j.ijrobp.2011.06.1679
- Aquino-Parsons C, Lomas S, Smith K, et al. Phase III study of silver leaf nylon dressing vs standard care for reduction of inframammary moist desquamation in patients undergoing adjuvant whole breast radiation therapy. J Med Imag Rad Sci.
2010;41(4):215–221. https://doi.org/10.1016/j.jmir.2010.08.005 - Zhong WH, Tang QF, Hu LY, et al. Mepilex Lite dressings for managing acute radiation dermatitis in nasopharyngeal carcinoma patients: a systematic controlled clinical trial. Med Oncol. 2013;30(4):761. https://doi.org/10.1007/s12032-013-0761-y
- Robijns J, Censabella S, Claes S, et al. Prevention of acute radiodermatitis by photobiomodulation: a randomized, placebo-controlled trial in breast cancer patients (TRANSDERMIS trial). Lasers Surg Med. 2018. https://doi.org/10.1002/lsm.22804. Epub ahead of print.
- Haruna F, Lipsett A, Marignol L. Topical management of acute radiation dermatitis in breast cancer patients: a systematic review and meta-analysis. Anticancer Res. 2017;37(10):5343–5353. https://doi.org/10.21873/anticanres.11960
- Ho AY, Olm-Shipman M, Zhang Z, et al. A randomized trial of mometasone furoate 0.1% to reduce high-grade acute radiation dermatitis in breast cancer patients receiving postmastectomy radiation. Int J Radiat Oncol Biol Phys. 2018;101(2): 325–333.
https://doi.org/10.1016/j.ijrobp.2018.02.006 - Zenda S, Yamaguchi T, Yokota T, et al. Topical steroid versus placebo for the prevention of radiation dermatitis in head and neck cancer patients receiving chemoradiotherapy: the study protocol of J-SUPPORT 1602 (TOPICS study), a randomized doubleblinded phase 3 trial. BMC Cancer. 2018;18(1):873. https://doi.org/10.1186/s12885-018-4763-1
- Liao Y, Feng G, Dai T, et al. Randomized, self-controlled, prospective assessment of the efficacy of mometasone furoate local application in reducing acute radiation dermatitis in patients with head and neck squamous cell carcinomas. Medicine (Baltimore). 2019;98(52):e18230. https://doi.org/10.1097/MD.0000000000018230
- Papier A, Strowd LC. Atopic dermatitis: a review of topical nonsteroid therapy. Drugs Context. 2018;7:212521.
https://doi.org/10.7573/dic.212521 - Jowkar F, Namazi MR. Statins in dermatology. Int J Dermatol. 2010;49(11):1235–1243. https://doi.org/10.1111/j.1365-4632.2010.04579.x
- Ghasemi A, Ghashghai Z, Akbari J, et al. Topical atorvastatin 1% for prevention of skin toxicity in patients receiving radiation therapy for breast cancer: a randomized, double-blind, placebo-controlled trial. Eur J Clin Pharmacol. 2019;6:1–8.
https://doi.org/10.1007/s00228-018-2570-x - Rahimi R, Abdollahi M. A systematic review of the topical drugs for post hemorrhoidectomy pain. Int J Pharmacol. 2012;8:628–637.
https://doi.org/10.3923/ijp.2012.628.637 - Wells M, Macmillan M, Raab G, et al. Does aqueous or sucralfate cream affect the severity of erythematous radiation skin reactions? A randomised controlled trial. Radiother Oncol. 2004;73(2):153–162. https://doi.org/10.1016/j.radonc.2004.07.032
- Falkowski S, Trouillas P, Duroux JL, Bonnetblanc JM, Clavere P. Radiodermatitis prevention with sucralfate in breast cancer: fundamental and clinical studies. Support Care Cancer. 2011;19(1):57–65. https://doi.org/10.1007/s00520-009-0788-y
- Elliott EA, Wright JR, Swann RS, et al. Phase III trial of an emulsion containing trolamine for the prevention of radiation dermatitis in patients with advanced squamous cell carcinoma of the head and neck: results of Radiation Therapy Oncology Group Trial 99-13. J Clin Oncol. 2006;24(13):2092–2097. https://doi.org/10.1200/JCO.2005.04.9148
- Fenig E, Brenner B, Katz A, et al. Topical Biafine and Lipiderm for the prevention of radiation dermatitis: a randomized prospective trial. Oncol Rep. 2001;8(2):305–309. https://doi.org/10.3892/or.8.2.305
- Gosselin TK, Schneider SM, Plambeck MA, Rowe K. A prospective randomized, placebo-controlled skin care study in women diagnosed with breast cancer undergoing radiation therapy. Oncol Nurs Forum. 2010;37(5). https://doi.org/10.1188/10.ONF.619-626
- Abbas H, Bensadoun RJ. Trolamine emulsion for the prevention of radiation dermatitis in patients with squamous cell carcinoma of the head and neck. Support Care Cancer. 2012;20(1):185–190. https://doi.org/10.1007/s00520-011-1110-3
- Shariati L, Amouheidari A, Naji Esfahani H, et al. Protective effects of doxepin cream on radiation dermatitis in breast cancer: A single arm double-blind randomized clinical trial. Br J Clin Pharmacol. 2020. https://doi.org/10.1111/bcp.14238
- Chan RJ, Blades R, Jones L, et al. A single-blind, randomised controlled trial of StrataXRT® – a silicone-based film-forming gel dressing for prophylaxis and management of radiation dermatitis in patients with head and neck cancer. Radiother Oncol. 2019;139:72‐78. https://doi.org/10.1016/j.radonc.2019.07.014
- Haubner F, Ohmann E, Pohl F, et al. Wound healing after radiation therapy: review of the literature. Radiat Oncol. 2012;7(1):162.
https://doi.org/10.1186/1748-717X-7-162 - Kang HC, Ahn SD, Choi DH, et al. The safety and efficacy of EGF-based cream for the prevention of radiotherapy-induced skin injury: results from a multicenter observational study. Rad Oncol J. 2014;32(3):156. https://doi.org/10.3857/roj.2014.32.3.156
- Kouvaris JR, Kouloulias VE, Plataniotis GA, et al. Topical granulocyte-macrophage colony-stimulating factor for radiation dermatitis of the vulva. Br J Dermatol. 2001;144:646–647. https://doi.org/10.1046/j.1365-2133.2001.04115.x
- Farrugia CE, Burke ES, Haley ME, et al. The use of aloe vera in cancer radiation: an updated comprehensive review. Complement Ther Clin Pract. 2019;35:126–130. https://doi.org/10.1016/j.ctcp.2019.01.013
- Heggie S, Bryant GP, Tripcony L, et al. A phase III study on the efficacy of topical aloe vera gel on irradiated breast tissue. Cancer Nurs. 2002;25(6):442–451. https://doi.org/10.1097/00002820-200212000-00007
- Atiyeh BS, Dham R, Kadry M, et al. Benefit-cost analysis of moist exposed burn ointment. Burns. 2002;28(7):659–663. https://doi.org/10.1016/S0305-4179(02)00075-X
- Geara FB, Eid T, Zouain N, et al. Randomized, prospective, open-label phase III trial comparing MEBO ointment with Biafine cream for the management of acute dermatitis during radiotherapy for breast cancer. Am J Clin Oncol. 2018;102(3):e610. https://doi.org/10.1016/j.ijrobp.2018.07.1672
- Gilca M, Tiplica GS, Salavastru CM. Traditional and ethnobotanical dermatology practices in Romania and other Eastern European countries. Clin Dermatol. 2018;36(3):338–352. https://doi.org/10.1016/j.clindermatol.2018.03.008
- Pommier P, Gomez F, Sunyach MP, et al. Phase III randomized trial of Calendula officinalis compared with trolamine for the prevention of acute dermatitis during irradiation for breast cancer. J Clin Oncol. 2004;22:1447–1453. https://doi.org/10.1200/JCO.2004.07.063
- Schneider F, Danski MT, Vayego SA. Usage of Calendula officinalis in the prevention and treatment of radiodermatitis: a randomized double-blind controlled clinical trial. Rev Esc Enferm USP. 2015;49(2):221–228. https://doi.org/10.1590/S0080-623420150000200006
- Friedrich EE, Sun LT, Natesan S, et al. Effects of hyaluronic acid conjugation on anti-TNF-α inhibition of inflammation in burns. J Biomed Mater Res A. 2014;102(5):1527–1536. https://doi.org/10.1002/jbm.a.34829
- Chen LH, Xue JF, Zheng ZY, et al. Hyaluronic acid, an efficient biomacromolecule for treatment of inflammatory skin and joint diseases: A review of recent developments and critical appraisal of preclinical and clinical investigations. Int J Biol Macromol. 2018;116:572–584. https://doi.org/10.1016/j.ijbiomac.2018.05.068
- Paganelli A, Mandel VD, Pellacani G, et al. Synergic effect of plasma exeresis and non-cross-linked low and high molecular weight hyaluronic acid to improve neck skin laxities. J Cosmet Dermatol. 2020;19(1):55–60. https://doi.org/10.1111/jocd.12976
- Gracy RLK, Phelps J. RadiaPlexRx Gel Protects Cultured Skin Cells from Oxidative Free Radical Damage Induced by Hydrogen Peroxide and by Irradiation: A Pilot Study. SSA20-08. Chicago, IL: Radiological Society of North America; 2005:300
- Liguori V, Guillemin C, Pesce GF, et al. Double-blind, randomized clinical study comparing hyaluronic acid cream to placebo in patients treated with radiotherapy. Radiother Oncol. 1997;42(2):155–161. https://doi.org/10.1016/S0167-8140(96)01882-8
- Cosentino D, Piro F. Hyaluronic acid for treatment of the radiation therapy side effects: a systematic review. Eur Rev Med Pharmacol Sci. 2018;22(21):7562–7572. https://doi.org/10.26355/eurrev_201811_16298
- Franco P, Potenza I, Moretto F, et al. Hypericum perforatum and neem oil for the management of acute skin toxicity in head and neck cancer patients undergoing radiation or chemo-radiation: a single-arm prospective observational study. Radiat Oncol. 2014;9:297. https://doi.org/10.1186/s13014-014-0297-0
- Huang CJ, Huang MY, Fang PT, et al. Randomized double-blind, placebo-controlled trial evaluating oral glutamine on radiation-induced oral mucositis and dermatitis in head and neck cancer patients. Am J Clin Nutr. 2019;109(3):606–614. doi:10.1093/ajcn/nqy329
- Scalia S, Marchetti N, Bianchi A. Comparative evaluation of different co-antioxidants on the photochemical- and functionalstability of epigallocatechin-3-gallate in topical creams exposed to simulated sunlight. Molecules. 2013;18:574–587. https://doi.org/10.3390/molecules18010574
- Zhao H, Zhu W, Jia L, et al. Phase I study of topical epigallocatechin-3-gallate (EGCG) in patients with breast cancer receiving adjuvant radiotherapy. Br J Radiol. 2015;89(1058):20150665. https://doi.org/10.1259/bjr.20150665
- Zhu W, Jia L, Chen G, et al. Epigallocatechin-3-gallate ameliorates radiation-induced acute skin damage in breast cancer patients undergoing adjuvant radiotherapy. Oncotarget. 2016;7(30):48607. https://doi.org/10.18632/oncotarget.9495
- Halperin EC, Perez CA, Brady LW. The discipline of radiation oncology. In: Halperin EC, Perez CA, Brady LW, eds. Principles and Practice of Radiation Oncology. Philadelphia, PA: Lippincott Williams & Wilkins; 2008:2–75.
- Reynolds JEF, ed. Martindale: The Extra Pharmacopoeia. 30th ed. Dexpanthenol. London: The Pharmaceutical Press; 1993:1361.
- Chan RJ, Mann J, Tripcony L, et al. Natural oil-based emulsion containing allantoin versus aqueous cream for managing radiationinduced
skin reactions in patients with cancer: a phase 3, double-blind, randomized, controlled trial. Int J Radiat Oncol Biol Phys. 2014;90(4):756–764. https://doi.org/10.1016/j.ijrobp.2014.06.034 - Manas A, Santolaya M, Ciapa VM, et al. Topical R1 and R2 prophylactic treatment of acute radiation dermatitis in squamous cell carcinoma of the head and neck and breast cancer patients treated with chemoradiotherapy. Eplasty. 2015;15:e25.
- Cui Z, Xin M, Yin H, et al. Topical use of olive oil preparation to prevent radiodermatitis: results of a prospective study in nasopharyngeal carcinoma patients. Int J Clin Exp Med. 2015;8(7):11000–11006.
- Palatty PL, Azmidah A, Rao S, et al. Topical application of a sandal wood oil and turmeric based cream prevents radiodermatitis in head and neck cancer patients undergoing external beam radiotherapy: a pilot study. Br J Radiol. 2014;87(1038):20130490. https://doi.org/10.1259/bjr.20130490
- Carrara M, Giandini T, Pariani C, et al. Comment on objective assessment in digital images of skin erythema caused by radiotherapy. Med Phys. 2016;43(5):2687. https://doi.org/10.1118/1.4945019
- Pazdrowski J, Polaſska A, Kaźmierska J, et al. Skin barrier function in patients under radiation therapy due to the head and neck cancers – preliminary study. Rep Pract Oncol Radiother. 2019;24(6):563–567.
- Salama SA, Arab HH, Omar HA, et al. L-carnitine mitigates UVA-induced skin tissue injury in rats through downregulation of oxidative stress, p38/c-Fos signaling, and the proinflammatory cytokines. Chem Biol Interact. 2018;285:40–47. https://doi.org/10.1016/j.cbi.2018.02.034
- Di Stefano G, Di Lionardo A, Galosi E, et al. Acetyl-L-carnitine in painful peripheral neuropathy: a systematic review. J Pain Res. 2019;12:1341–1351. https://doi.org/10.2147/JPR.S190231
- Giacomelli L, Moglia A, Losa G, Quaglino P. Clinical use of Capilen, a liposomal cream based on fresh plant extracts enriched with omega fatty acids. Drugs Context. 2020;9:2019-10-1. https://doi.org/10.7573/dic.2019-10-1
- Serrano G, Almudéver P, Serrano JM, et al. Phosphatidylcholine liposomes as carriers to improve topical ascorbic acid treatment of skin disorders. Clin Cosmet Investig Dermatol. 2015;8:591–599. https://doi.org/10.2147/CCID.S90781
- Brand RM, Epperly MW, Stottlemyer JM, et al. A topical mitochondria-targeted redox cycling nitroxide mitigates oxidative stress induced skin damage. J Invest Dermatol. 2017;137(3):576–586. https://doi.org/10.1016/j.jid.2016.09.033
- Imai T, Matsuura K, Asada Y, et al. Effect of HMB/Arg/Gln on the prevention of radiation dermatitis in head and neck cancer patients treated with concurrent chemoradiotherapy. Jpn J Clin Oncol. 2014;44(5):422–427. https://doi.org/10.1093/jjco/hyu027


